Founded by Christina Fullerton in 2021 while she was on maternity leave with her first born Daughter Annie, this gorgeous baby range contains essential products for any parent. The first must-have for any changing bag is Bunny & Birdy’s hypoallergenic & antibacterial muslin cloths. They are a 70% bamboo and 30% cotton blend, making them incredibly soft and very durable. They are perfect to use as a burping cloth, bib, changing mat or a washcloth at bath time. With so many uses this is the one item that should be in every changing bag.
The soother Clip is the next saviour product in the range. Keep those precious soothers at hands reach at all times. These Soother clips keep your babies best friend safe and easily located when out and about. The strap is made from 100% organic cotton, with a classic beech clip making it incredibly easy to use and most importantly, very durable.
The soother Clip is the next saviour product in the range. Keep those precious soothers at hands reach at all times. These Soother clips keep your babies best friend safe and easily located when out and about. The strap is made from 100% organic cotton, with a classic beech clip making it incredibly easy to use and most importantly, very durable.
Other products of note include the BPA free silicone soother pod, designed to keep baby's soother clean and safe when not being used. This airtight and easily cleaned pod can fit 2 soothers or a soother and a soother clip.
The milk powder dispenser is ideal for anyone who often forgets how many scoops of formula they've put into a container. The milk powder dispenser from Bunny & Birdy holds 150g and comes with a handy 5g scoop. The clear lid and scoop leveller are detachable so when baby has come off formula, it can be used as a snack pot for on-the-go.
Lastly, the super soft lightweight blanket from Bunny & Birdy is the perfect size and thickness to keep little one's cosy and comfortable. With fun new patterns coming soon to the blanket range, this is a brand you'll want to keep your eyes on.
You can check out the full product listing at the link below:
https://bit.ly/45PgYaa
Instagram:
https://www.instagram.com/bunny_andbirdy/
You can check out the full product listing at the link below:
https://bit.ly/45PgYaa
Instagram:
https://www.instagram.com/bunny_andbirdy/
PSL are immensely proud to be the official distributor for Green Angel - Luxury Irish Skincare.
Green Angel was founded in 2006 by Chris and Mary Mitchell. They saw a gap in the market for a natural skincare brand that really works. From a young age, Mary was aware of the extraordinary healing and therapeutic benefits of natural plants oils. She went on a journey into the amazing world of magical botanical energy and infinitely effective ingredients and from there Green Angel was born.
They attended tradeshows up and down the Isle of Ireland, the products spoke from themselves as their reputation and popularity grew every year. As the brand expanded, they brought Green Angel overseas to tradeshows in the UK and Europe. It was a massive success and Green Angel now delivers a piece of Ireland to addresses worldwide.
Green Angel are extremely passionate about their products which have a holistic approach that leaves the whole body revitalised and enriched. The luxurious and natural range use only the finest ingredients, harnessing the power of hand-harvested organic Irish seaweed from the West Coast of Ireland.
They produce a range of products from diffusers, candles and room sprays to face creams, body lotions, shampoos and conditioners. The skincare and body care products are sold in a range of beautifully presented gift sets, perfect to give as presents at any time of the year.
You can check out their full range of products on the PSL website here: https://bit.ly/44Fsy6o
For further information on the brand, you can also check them out directly here: https://www.greenangel.com
Instagram: https://www.instagram.com/greenangelskin/
Facebook: https://www.facebook.com/GreenAngelSkincareProducts
Facebook: https://www.facebook.com/GreenAngelSkincareProducts
As a rooted Irish health and beauty brand, Pharmacy Supplies Limited (PSL) was established about 30 years ago with the aim of providing service with quality, ethical values and a diverse product range. PSL works with Brand Partners by offering bespoke dynamic sales and marketing solutions and fitted distribution strategies to contribute to brands' growth and build trust between them. The target sectors of PSL are pharmacies, multi-national grocery, department stores and online retailers.
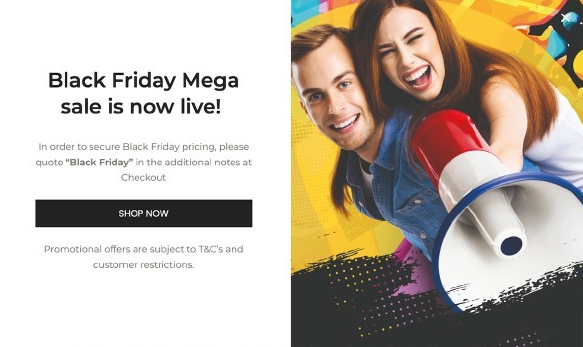
While connecting the right brands with the right retailers, PSL uses Popupsmart to increase engagement and interaction for their campaigns. The main goal of using Popupsmart was to engage with customers and increase the conversion rate most effectively at the same time. Appealing to the direct needs of its brands, PSL used the right segmentation and integration options to keep track of its audience. By creating diverse pop up campaigns for the Black Friday period, PSL strategically attracted and met the needs of its brand partners with creative pop ups.



While connecting the right brands with the right retailers, PSL uses Popupsmart to increase engagement and interaction for their campaigns. The main goal of using Popupsmart was to engage with customers and increase the conversion rate most effectively at the same time. Appealing to the direct needs of its brands, PSL used the right segmentation and integration options to keep track of its audience. By creating diverse pop up campaigns for the Black Friday period, PSL strategically attracted and met the needs of its brand partners with creative pop ups.

By using a related image to announce the Black Friday mega sale to customers with a legible header, eye-catching text and a CTA button that leads to a specific page, they grabbed the attention of website visitors. PSL also used geolocation segmentation in order not to appear in the USA and targeted every session for a one-time display to provide a seamless experience. With the Google Analytics 4 integration, PSL was able to follow the analytics on GA4 along with the Leads & Analytics pages of Popupsmart.
The results of the campaign showed it was hugely successful with a 6.44% conversion rate, 419 interactions and 6,505 pageviews.
Not only did PSL use Popupsmart for their Black Friday promotions, they also use Popupsmart regularly for weekly offer pop-ups on their site. The promo offers popup of PSL stands out with its mobile view, and it has the right proportion of image, headline, text and button. Displaying the popup for one week, PSL made use of the GA4 integration to keep tracks of its impact on the Leads & Analytics pages of Popupsmart. PSL reached 5,346 pageviews with a conversion rate of 4.94% by interacting with its visitors only for a charming and informative weekly offer.

PSL's review of Popupsmart:
"Popupsmart's easy-to-use interface made it simple for us to create successful campaigns. The popups caught the attention for our promotions, especially during Black Friday, and helped promote our weekly offers. The quick and efficient support from the Popupsmart team made our experience a success and we will be continuing to use their services throughout 2024."
Sarah Keary is one of Ireland's leading make-up artists and the creative force behind By SK. Her journey in the cosmetics industry began in 2004, and she has since solidified her reputation as a true makeup master. Over the years, Sarah's artistry has graced the faces of some of Ireland's leading media personalities, her talent and dedication to her craft quickly propelled her to the forefront of the industry, where she has become a trusted name.
The By SK makeup range has something for everyone from lipstick, lip liner and lip gloss to brow pencils, eye pencils and liquid blushers.
The very popular 'Palette of Dreams' is a versatile and instant full face luxury palette with a richly pigmented, long wearing and easy to blend formula ensuring minimal fall down or caking. It includes bronzer, highlighter, blusher and 6 eyeshadows so it's perfect for taking a look from day to night or for everyday wear.
By SK also has a range of brush sets available with the 5 piece velvet brush set being the most popular. The 5 piece set includes a foundation brush, powder/blusher brush, eye duo brush, eyeshadow/concealer brush and a liner/brow brush. The velvet bag is also included. Sarah Keary notes, "These are my absolute go-to brushes for a quick on-the-go look. Every piece is versatile and easy to use."
Lastly, of note, is the By SK radiant primer. Another best seller, this ultra-lightweight primer glides onto the skin effortlessly leaving a silky-smooth canvas ready for make up application. The 100% vegan formula has anti-ageing skin benefits and helps to soothe and repair the skin.
You can check out the full product listing at the link below
https://bit.ly/45YiLJy
For more information on Sarah Keary, check out the website below:
https://sarahkeary.ie/
Instagram:
https://www.instagram.com/sarahkearymakeup/
Facebook:
https://www.facebook.com/sarahkearymakeup
You can check out the full product listing at the link below
https://bit.ly/45YiLJy
For more information on Sarah Keary, check out the website below:
https://sarahkeary.ie/
Instagram:
https://www.instagram.com/sarahkearymakeup/
Facebook:
https://www.facebook.com/sarahkearymakeup
PSL are the official distributor of the fabulous Sorted Skin - no-nonsense skincare brand.
There are millions of little helpers looking out for you right now! Good bacteria lives on the surface of your skin and protects you 24/7, promoting repair and keeping you safe from infection. When this good bacteria is overpowered by bad, it can lead to conditions like eczema, acne and rosacea.
Sorted Skin is packed with powerful prebiotics, which feeds the skin's good bacteria so it can keep battling against the bad! The no-nonsense, microbiome-based skincare brand that does things differently, understanding how having problematic skin may affect your daily life. They put a huge emphasis on health over looks. Believing in equipping the skin with exactly what it needs to heal and repair itself. That's why their strapline is 'A Balanced Diet for Your Skin'. They put skin health first, not skin beauty, because healthy skin is beautiful skin.
The best-selling 5 in 1 Anti-Redness Day Cream with SPF 50 gives an instant solution to the appearance of red, sensitive skin and is suitable for use by people who suffer from rosacea, blemishes and spots, flushing, spider veins and other causes of redness on the skin.
The Intensive Rescue range should be noted as well. The range delivers moisture-locking technology for up to 72 hours to rejuvenate, rebalance and restore skin. The range is suitable for people prone to eczema, dermatitis, psoriasis, dry dehydrated skin and allergic reactions such as itching.
The Intensive Rescue Spray is a fast-acting liquid spray which offers instant hydration and relief from the symptoms of dry atopic and sensitive skin. The spray also won the Editor's Choice Gold Award at the Global Green Beauty Awards 2023 being particularly commended for the quick relief it delivers to itchy and dry skin.
The Intense Rescue Moisturiser targets extremely dry, cracked and damaged skin on the body to relieve irritation, and provide intense nourishment, moisturisation and protection to the surface layers of the skin. The moisturiser also won the Bronze Award for the Best Natural Skincare Product at the Global Green Beauty Awards 2023, being particularly commended for the repairing, rejuvenation and restoration of very dry, cracked and damage skin.
Sorted Skin is 100% natural, vegan and suitable for many different skin types.
You can check out their full range of products here: https://bit.ly/3qWGs6n
For more information on the brand, check out their website at the following link: https://sortedskin.com/
Instagram - https://www.instagram.com/sorted.skin/
Facebook - https://www.facebook.com/Sorted.Skin
For more information on the brand, check out their website at the following link: https://sortedskin.com/
Instagram - https://www.instagram.com/sorted.skin/
Facebook - https://www.facebook.com/Sorted.Skin
The Goat Skincare story isn't just about a product, or a magic ingredient...it's about family. And how wanting the best for their family led them to create a product that could be loved and enjoyed by families across the world.
They believe that traditional, natural remedies are the best. Gentle hydration for dry and sensitive skin using simple, natural ingredients and the power of pure, ethically-sourced goat's milk, they have created a range that will nurture the skin of your entire family - babies included.
Products are made using the key ingredient of pure goat's milk which is ethically farmed and sourced in Victoria. They're free of parabens, sulphates, fragrances and colourants and are pH balanced so they can be used daily, without irritating the skin.
The Goat Skincare range is suitable for all ages and all skin types. And, because we wouldn't be here without them, they don't ever test on animals.
Renowned for its soothing properties, it's packed with vitamins and contains essential fatty acids which make it ultra moisturising for optimal skin suppleness. Product lines include body wash, shampoo and conditioner , moisturising cream, moisturising lotion, soap bars and the best-selling baby goat range.
The Baby Goat Skincare has been specifically designed to care for the delicate skin of new borns and infants. Helping to soothe redness, itching and inflammation.
Check out The Goat Skincare on the PSL website: https://bit.ly/3PriTMk
For further information on the brand, you can also check them out directly via their website: https://thegoatskincare.com.au
Instagram: https://www.instagram.com/thegoatskincare/
Facebook: https://www.facebook.com/thegoatskincare
Tiktok: https://www.tiktok.com/@thegoatskincare
Facebook: https://www.facebook.com/thegoatskincare
Tiktok: https://www.tiktok.com/@thegoatskincare





